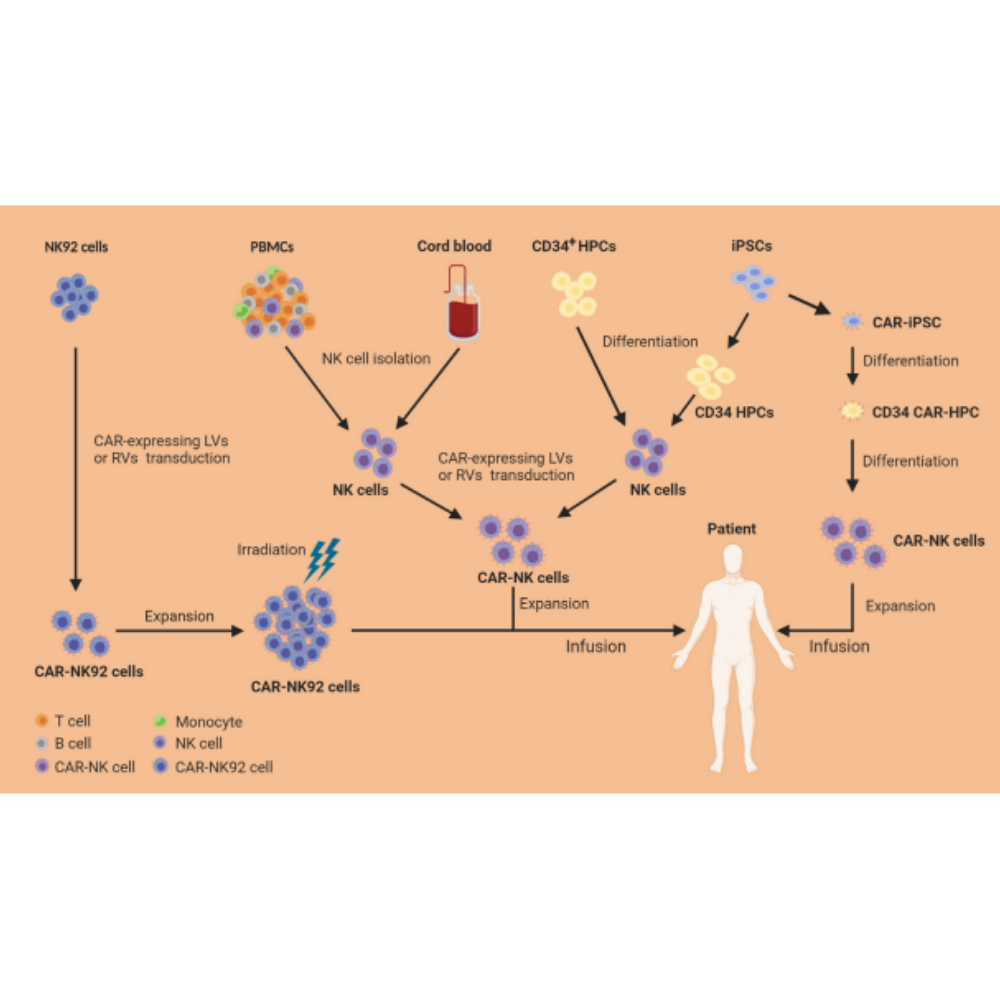
Natural Killer (NK) cells and CD8+ cytotoxic T cells are two types of immune cells that can kill target cells through similar cytotoxic mechanisms. With the remarkable success of chimeric antigen receptor (CAR)- engineered T (CAR-T) cells for treating haematological malignancies, there is a rapid growing interest in developing CAR-engineered NK (CAR-NK) cells for cancer therapy. Compared to CAR-T cells, CAR-NK cells could offer some significant advantages, including: (1) better safety, such as a lack or minimal cytokine release syndrome and neurotoxicity in autologous setting and graft-versus-host disease in allogenic setting, (2) multiple mechanisms for activating cytotoxic activity, and (3) high feasibility for ‘off-the-shelf’ manufacturing.
In this review, we focus on recent progress in genetic engineering and clinical application of CAR-NK cells, and discuss current challenges and future promise of CAR-NK cells as a novel cellular immunotherapy in cancer.
CAR-NK cells have a great promise as a novel cellular immuno- therapy platform against cancer and with a potential for generating “off-the-shelf” products that could be readily available and safe for clinical use.
Recent advances in gene manipulation techniques have allowed for the creation of novel CAR-NK cell products with potent anti-tumour activity but mostly non-toxic to normal tissues after infusion into the patients. Multiple strategies, including CRISPR based genetic modifications and inclusion of novel genes to modulate TME in the CAR constructs are some of the key innovations helping to sig- nificantly advance the field. With the increasing safety and promising activity in preclinical studies and clinical trials, combined with advanced efforts addressing the remaining challenges, it is expected that CAR-NK cell therapy will continue to evolve and lead to major improvement in the survival of cancer patients with otherwise very limited treatment options.
Article Reference link: click here
Scientific article publishing date: 19/09/2020
Article identifier BCN22_411EN

The foremost Cancer Therapy is here.
;
;